An antisense RNA expression vector for Neurospora crassa
W. Fecke, U. Nehls and H. Weiss - Institut für Biochemie, Heinrich Heine Universität
Düsseldorf, Universitätsstrasse 1, 4000 Düsseldorf 1, Federal Republic of Germany
The artificial expression of antisense RNA is commonly used in eucaryotes,
especially higher plants, to reduce the level of specific proteins (van der Krol et al. 1988
Nature 333:866-869). Here we report the use of antisense RNA to inhibit the translation
of a subunit of the mitochondrial NADH:ubiquinone oxidoreductase, the respiratory
complex I in N. crassa.
Fragments from the quinic acid inducible qa gene cluster (Geever et al. 1989 J.
Mol. Biol. 207:15-34) were used to construct an antisense RNA expression vector (Figure
1). A full-length cDNA of the 51 kDa NADH binding subunit of complex I (Weiss et
al. 1991 Eur. J. Biochem. 197:563-576) was inserted in reverse orientation between a
promoter frag-ment of the qa-2 gene, including four regulation sites, and a
polyadenylation and termination fragment of the qa-4 gene. The am gene was used as a
selectable marker for transformation of am deficient mutants (Kinsey and Rambosek
1984 Mol. Cell. Biol. 4:117-122)
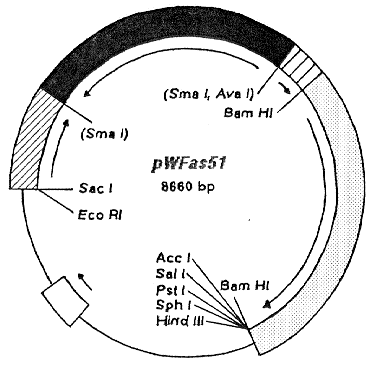
Figure 1. Construction of the antisense RNA expression vector pWAFas51. A 380 bp BamHI/AvaI
fragment of the qa-4 gene, which includes regions for polyadenylation and termination of the qa-4 RNA, was
inserted into the multiple cloning site of the parental pT7T3 (Pharmacia) by destroying the AvaI site. A 980
bp SacI/SmaI fragment of the qa-2 gene with the promoter region and four regulation sites - for induction
with quinic acid - was also inserted in the MCS. A 2600 bp BamHI fragment of the am gene was used as a
selectable marker and ligated into the BamHI site of the vector. The recessed 3' ends of the 1800 bp cDNA
of the 51 kDa gene were filled in and the blunt-ended molecules were ligated in reverse orientation into the
SmaI site of the RNA expression vector; the SmaI site was destroyed by this step. The open box represents
the ampicillin resistance gene while the dark-slashed and light-slashed boxes indicate the qa-2 and qa-4 areas
respectively. The filled box represents the cDNA of the 51 kDa gene and the stippled box shows the am
gene. The arrows indicate the direction of transcription.
After transformation, the integration of the plasmid into the genome was detected
by Southern blot analysis. In heterokaryotic transformants grown with quinic acid as sole
carbon source, the antisense RNA was expressed (Figure 2) leading to 50 to 80%
reduction of the translation of the subunit (Figure 3), depending on the time of
incubation with quinic acid. No difference in the level of transcript from the endogenous
51 kDa gene could be ascertained between transformants grown with quinic acid nor with
sucrose as sole carbon source. Transformants grown with sucrose showed no reduction
of the 51 kDa protein and only a slight expression of antisense RNA. The transformants
showed no defects in mitochondrial respiration (see also Nehls et al. 1992 J. Mol. Biol.
227:1032-1042).
The described technique is superior to other methods (such as gene disruption)
whenever the complete loss of a protein has lethal consequences for the organism. The
inducible RNA expression vector without the am gene and the 51 kDa gene (named
pWF1) is available in the Fungal Genetics Stock Center. Each cDNA to be transcribed
can be blunt-ligated into the SmaI site of the vector, in sense or antisense orientation.
The unique restriction sites EcoRI, BamHI, AccI, SalI, PstI, SphI and HindIII can be
used for ligating different N. crassa selectable transformation markers into the vector.
The financial support of the Deutsche Forschungsgemeinschaft is acknowledged.
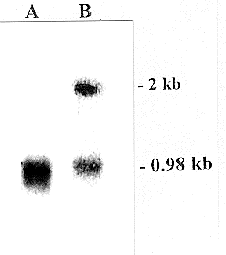
Figure 2. Northern blot analysis of antisense RNA expression in mutant (lane B) and wild type (lane A)
cells. Total RNA was isolated, subjected to electrophoresis in a formaldehyde agarose gel and transferred to
a nitrocellulose membrane. The RNA was hybridized with in vitro labelled transcripts from the 51 kDa gene
(in the sense orientation). As a control, an in vitro labelled antisense transcript from the 21.3 kDa gene,
which encodes a subunit of the membrane arm of complex I, was included in the hybridization. A signal
with the expected 2 kb length of the antisense RNA for the 51 kDa protein could be seen only in the
transformant RNA (lane B), while both wild type and transformant RNAs have a 0.98 kb transcript which
hybridized with the control probes for the 21.3 kDa protein.

Figure 3. Western blot analysis of one mutant (lane B) compared with the wild type (lane A). The same
amount of mitochondrial protein was incubated with antisera against the 51 kDa subunit and, as a control,
against the 21.3 kDa subunit of the membrane arm of complex I. The level of 51 kDa protein in the mutant
is reduced by 80% compared to the wild type.


